Area doctors join Lyme disease vaccine study, participants sought
news@greenecountymessenger.com
Two Uniontown doctors are participating in Lyme disease research that could result in a vaccine to prevent the tick-borne illness.
Dr. Richard Cook II and Dr. Paul Hartley, who have separate practices but both work within the Preferred Primary Care network, have been asked to join the study that is currently taking place in the U.S. and Europe.
“We were selected to participate because Lyme disease is so common in our area of Western Pennsylvania,” Cook said. “Our area was actually chosen as one of the bigger places to conduct the research.”
Pennsylvania as a whole reported the highest number of Lyme disease cases in the U.S. in 2019, according to the Centers for Disease Control and Prevention. There were 6,763 confirmed cases, and 2,235 probable cases reported that year. The second highest in the U.S. was New York, with 2,847 confirmed cases and 1,396 probable cases.
Cook and Hartley, who was not available for comment, will be administering three sets of vaccines, monitoring the results and working with Pfizer to provide the pharmaceutical company with information.
Lyme is a growing problem, with cases rising and warming weather helping ticks expand their habitat. While a vaccine for dogs has long been available, the only Lyme vaccine for humans was pulled off the U.S. market in 2002 from lack of demand, leaving people to rely on bug spray and tick checks.
“There wasn’t such a recognition, I think, of the severity of Lyme disease” and how many people it affects the last time around, Pfizer vaccine chief Annaliesa Anderson told The Associated Press.
According to the CDC, Lyme disease is transmitted to humans through the bite of infected blacklegged ticks. Symptoms include fever, headache, fatigue and a skin rash.
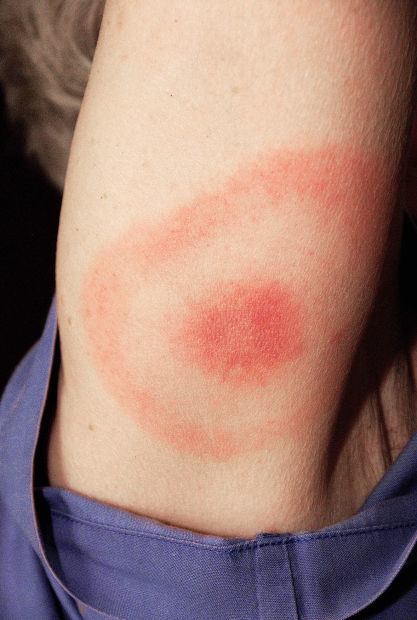
The most common sign of Lyme disease is a rash that looks like a bullƒ¢πΩ ”∆µ eye, according to Cook.
‚ÄúThe bullƒ¢πΩ ”∆µ-eye rash has a red ring, and then normal tissue and then a red ring again,‚Äù he said. ‚ÄúThatƒ¢πΩ ”∆µ a very good sign that you have Lyme disease.‚Äù
Although some people who contract Lyme disease do not have the rash, Cook said they will often complain of flu-like symptoms. They will also be very tired and achy.
If left untreated, Cook said the infection can spread to the joints, the heart and the nervous system.
“People who are afraid that they may have contracted the disease or people who work outside in the woods or do a lot of hiking through the woods are at risk,” Cook said. “They can take a blood test to find out what the results are.”
Cook said the enrollment period for Lyme disease patients will continue until Pfizer receives the numbers that it needs to complete the study. After the trials are concluded, participants will continue to be checked to determine if they have immunity for two years, Cook said.
The potential vaccine targets an ‚Äúouter surface protein‚Äù of the Lyme bacterium called OspA thatƒ¢πΩ ”∆µ present in the tickƒ¢πΩ ”∆µ gut. Itƒ¢πΩ ”∆µ estimated a tick must feed on someone for about 36 hours before the bacteria spreads to its victim. That delay gives time for antibodies the tick ingests from a vaccinated personƒ¢πΩ ”∆µ blood to attack the germs right at the source.
In small, early-stage studies, Pfizer and Valneva, a French biotech company, reported no safety problems and a good immune response. The newest study will test if the vaccine, called VLA15, really protects and is safe. The companies aim to recruit at least 6,000 people in Lyme-prone areas including the Northeast U.S. plus Finland, Germany, the Netherlands, Poland and Sweden.
‚ÄúWe‚Äôre really looking at something thatƒ¢πΩ ”∆µ a seasonal vaccine,‚Äù Anderson said, so people have high antibody levels during the months when ticks are most active.
Volunteers can be as young as 5 and should be at high risk because they spend a lot of time in tick-infested areas.
Those interested in participating in the vaccine trial can call Cookƒ¢πΩ ”∆µ office at 724-438-1479 or Hartleyƒ¢πΩ ”∆µ office at 724-437-1109
The Associated Press contributed to this report.